 Oxygen atom Oxide ion O O2- Negative ions end their name in ide. The ions attract each other and stick together.Ħ Li+ Li O2- O ION SYMBOLS Lithium atom Lithium ion When an atom gains an electron it becomes a negatively charged ion. 
When an atom loses an electron it becomes a positively charged ion. Charged atoms are called IONS.ĥ Atoms have equal numbers of positive charges (protons) in the nucleus and negative electrons.Ītoms are NEUTRAL. When an electron transfers from one atom to another the atoms will no longer be electrically neutral. One way atoms can join is to TRANSFER one or more electron(s) from one atom to another. It is the outer electrons on an atom which gives the atom its chemical properties. The numbered columns are called GROUPS.Ĥ ATOMS JOINING Atoms use their outer electrons to join. The energy levels further out from the nucleus can hold increasing numbers of electrons. There are 7 energy levels in an atom and there are 7 periods (rows). The periods relate to the filling up of the energy levels with electrons. The rows of the Periodic Table are called the PERIODS. Work out the number of protons, electrons and neutrons in an ion, given the mass number, atomic number and charge on an ion. State that an ion can be made from an atom by losing or gaining one or more electrons. 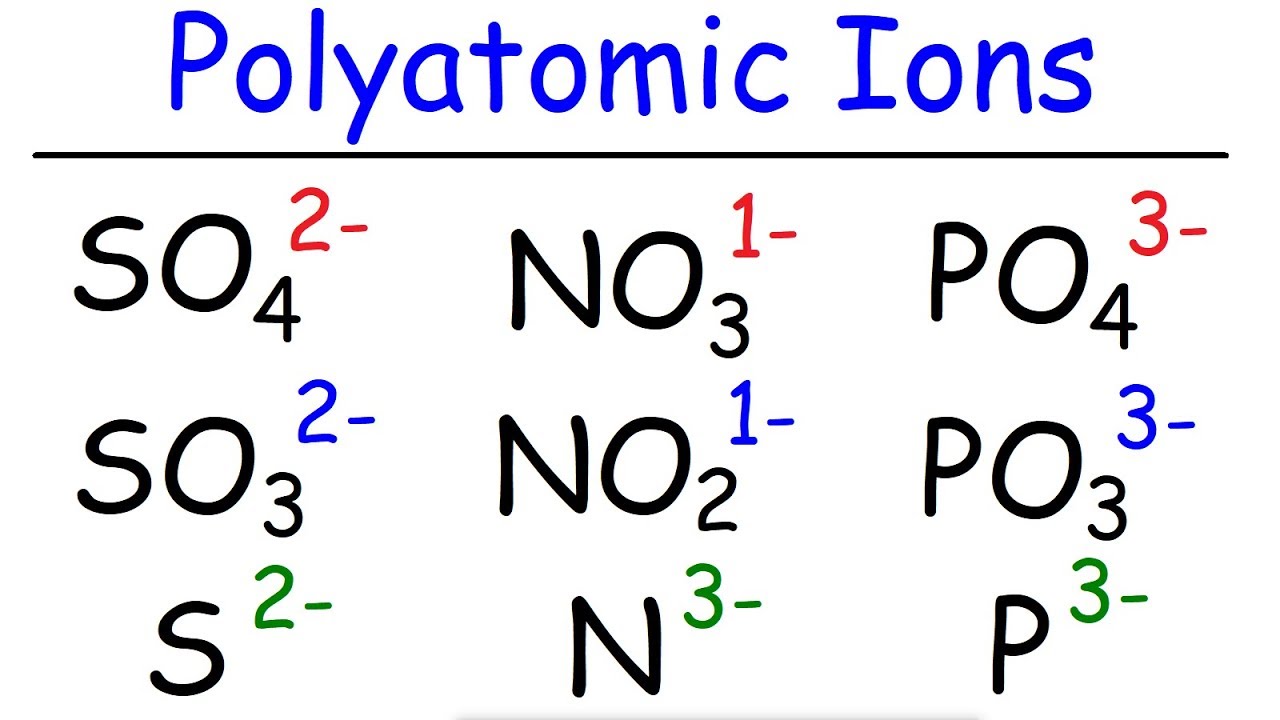
State that an ion is an atom which has a charge. State what period and a group are in the periodic table. CHEMISTRY N5 CHEMISTRY ATOMIC STRUCTURE THE PERIODIC TABLE & IONSĬHEMISTRY N5 CHEMISTRY ATOMIC STRUCTURE THE PERIODIC TABLE & IONS After completing this topic you should be able to : Relate the position of an element in the periodic table to the arrangement of its electrons in the atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
